  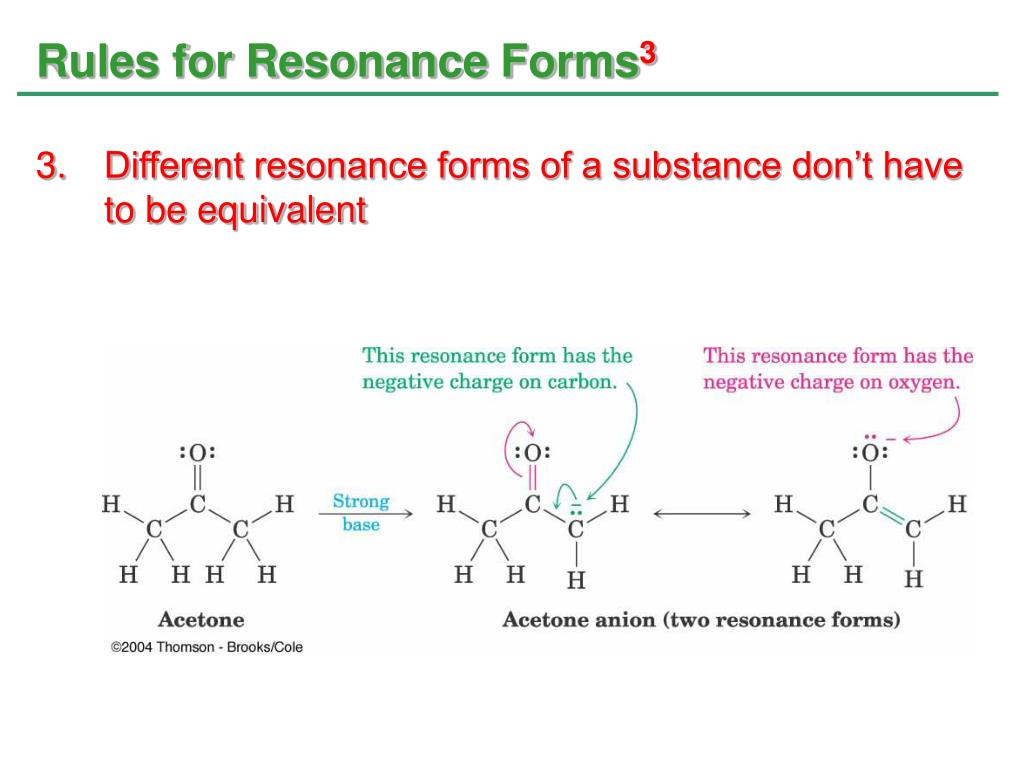
The actual structure is for the most part in between with the hydrogen floating between the carbon and the oxygen and the double bond spread out between the C-C and the C-O bonds. The same kind of Lewis structure wave function idea can be applied to resonance forms (which is what Pauling intended). 2: Resonance structures for keton/enol.Īs seen, the hydrogen marked with blue can be found next to the carbonyl group, which is the traditional 2D way of drawing a keton, but the hydrogen can also move over to the oxygen, forcing the formation of an alcohol group and a C-C double bond. 2) are of importance, as the stability of the isomers affect the efficiency of the reaction in various addition and elimination reactions.įig. The resonance structures are important, as they explain several effects in organic synthesis like why the benzene structure is so stable and why some substitutions like amine on benzene directs ortho and para (positions 2, 4, and 6) while others like the nitro group direct meta (positions 3 and 5) in electrophilic aromatic substitution.Įspecially the keton/enol isomers (fig. What is the practical importance of resonance structures? 
The -bondsin double or triple bonds can move without destroying the connectivity, since the -bond remains untouched. Therefore, such electrons are rarely moved. The difference from rearranging the electrons is called resonance structures, and the change between structures is shown by a single arrow pointing both ways. Sigma bondsare the strongest type of bond and represent the glue that holds the atoms together. The bond is said to be delocalized, which is why the benzene ring is often written as a hexagon with a circle. The double bond between position 1 and 2 is there some of the time, but on average there is really 1.5 bonds between all the carbons. When drawing the ring, the double bonds can be located in 2 different positions as shown in fig. Benzene is a ring of 6 carbon atoms with conjugated double bonds (i.e. The classical example in resonance structures is the benzene ring. Resonance - Electron Delocalization In certain types of organic molecule electrons are able to spread out over multiple atoms in order to stabilize the overall. In practical chemistry and advanced descriptions, resonance structures go a long way in describing chemical reactions or lack thereof. It is not possible to determine the contribution from each vibration because of the resulting mixed wave function.Usually we think of bonds as localized or static structures, and in most cases they act like they are, at least for basic descriptive purposes. An interaction of an empty p-orbital and. As a result, two strong bands are observed in the spectrum, instead of the expected strong and weak bands. There are 4 types of resonance that youll have to know within the scope of your organic chemistry course. The wavefunctions for the two resonant vibrations mix according to the harmonic oscillator approximation, and the result is a shift in frequency and a change in intensity in the spectrum. The two bands are usually a fundamental vibration and either an overtone or combination band. \left ( 2 \right ) \] Figure 4: Example of hot bands in a vibrational line spectrum of a diatomic molecule: (A) harmonic frequencies (B) hot band transitions (C) combination of both spectra.įermi resonance results in the splitting of two vibrational bands that have nearly the same energy and symmetry in both IR and Raman spectroscopies. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
